
CLINICAL STUDIES

OVERVIEW
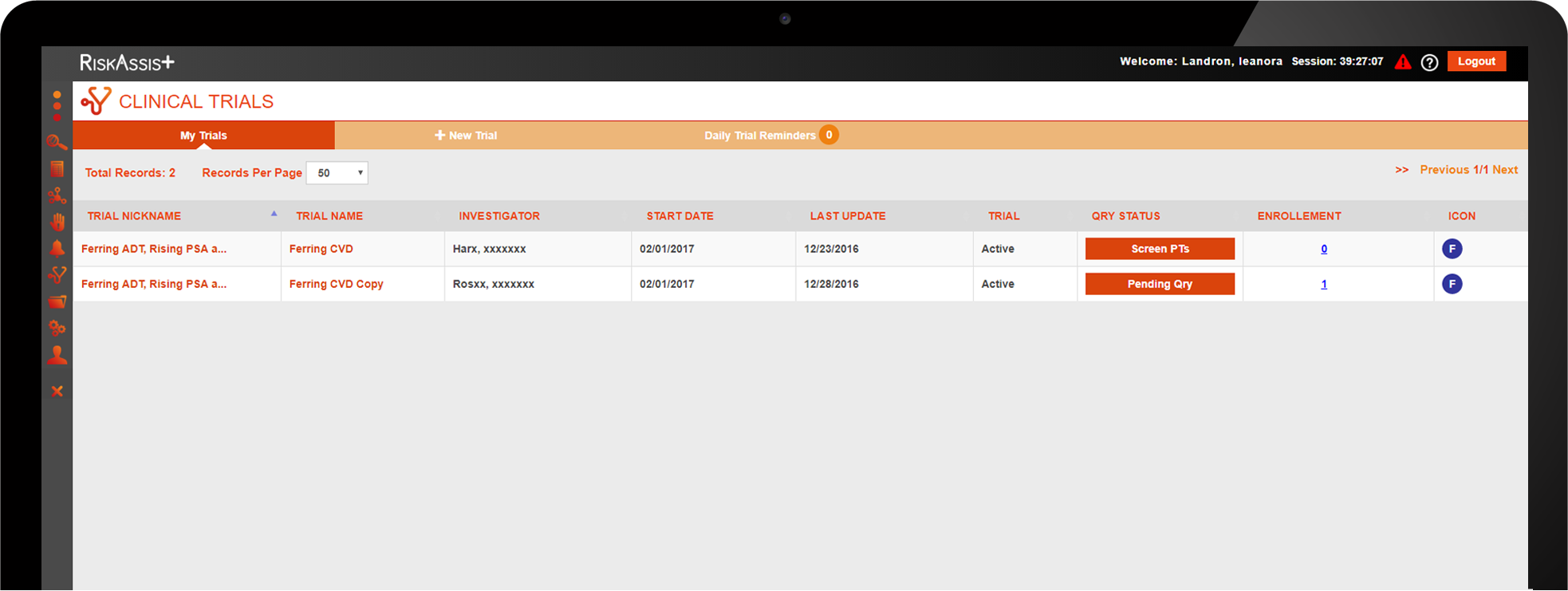
Clinical studies software from RiskAssist makes it easy for any clinic to perform clinical trials when they need to or want to and without the uphill battle of the paperwork.
Problem: Clinics leave revenue on the table by passing on studies because they cumbersome to administer, require too much paperwork and staff time, and clinics don’t have the tools to efficiently qualify, enroll, manage study patients.
Solution: Our clinical trials management solution manages the paperwork on the back end, requiring only minimal staff resources.
Easily administer trials and maximize revenue.

HIGHLIGHTS
- Automated case identification
- Effortless compliance
- Evidence-based
- Use national guidelines
- Or develop your own
- Program your own prompts and triggers
- Case management based on your workflow
- Aligned to your specialty and clini

BENEFITS
- Professional Development; Remain at the cutting edge of your specific area of therapeutic interest
- Professional Recognition; Clinical trial investigators are often recognized as thought leaders within the profession.
- Access to promising new options often not available outside the trial setting
- Patient monitoring, advice, care, and support by a research team (CRO)
- Opportunity for your patients to be the first to benefit from a new method
- Contributing to scientific knowledge
- New Revenue Stream. Clinical trials offer the opportunity for additional revenue.
- Role in the Evolution of Medicine. Clinical trial investigators can potentially bring breakthrough products to the market





